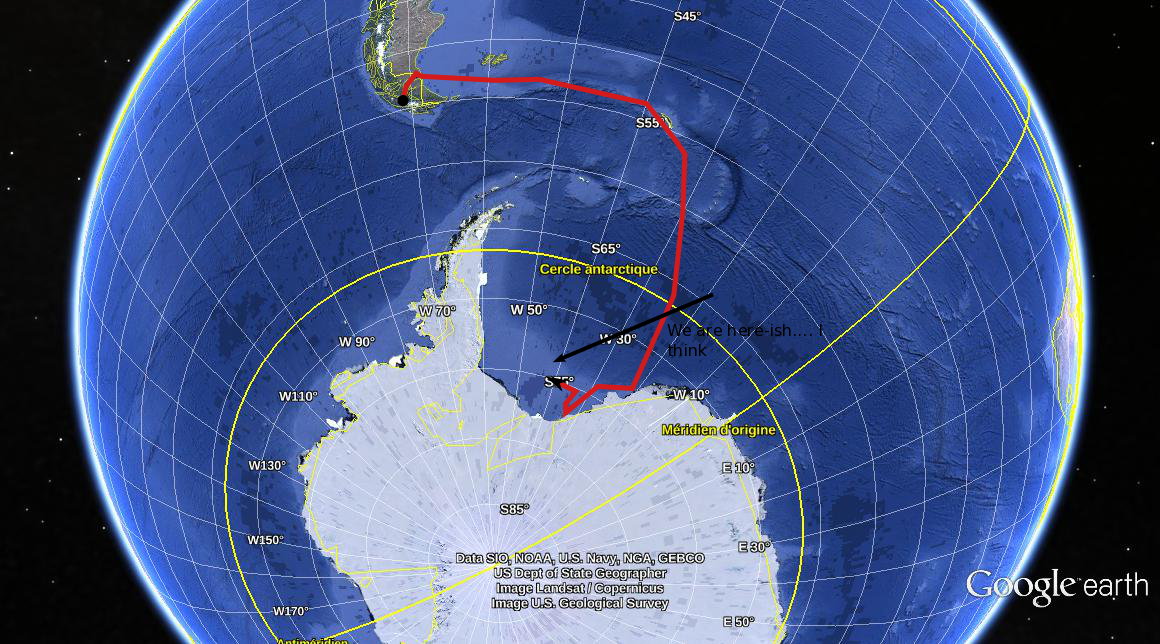
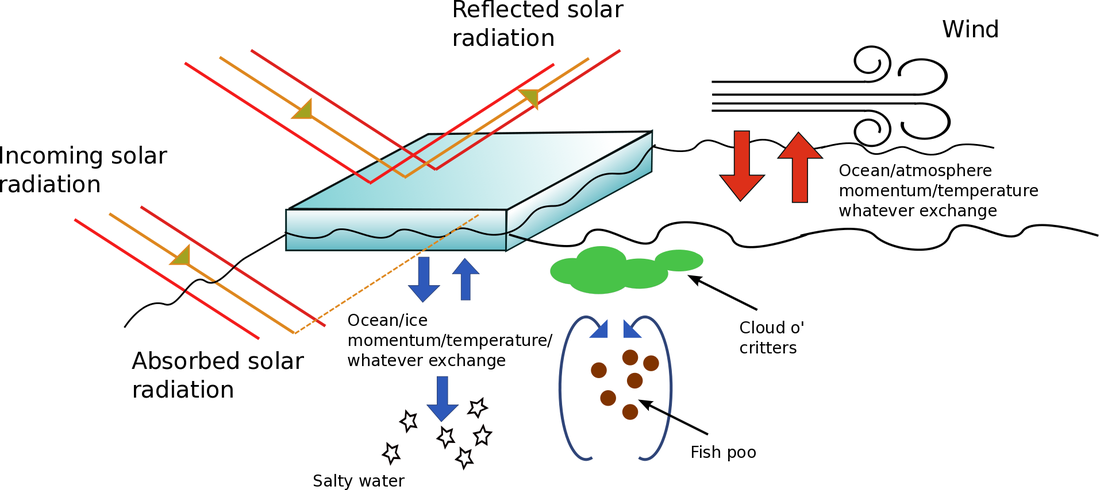
 I'm sorry. I'm afraid that there's no more room. You'll have to find another... I'm sorry. I'm afraid that there's no more room. You'll have to find another... I had originally planned to write another post on oceanographic instrumentation, in particular, Lagrangian drifters. But recently, we aboard the James-Clarke Ross have managed to get into the very far south-eastern part of the Weddell Sea where we've become surrounded by ice. Sea ice in particular. We're not stuck (well not yet), but ice conditions have degraded over the last couple of days, which may hamper our ability to go where we want to go. So, given that I've been staring at the stuff for the last few days, I thought I'd take the opportunity to kick off what will hopefully be a multi-part post on sea ice - what it is, why it's there, and why it's important to know about. It's also a good opportunity to post a bunch of photos. Lagrangian drifters will just have to wait. So, without further ado, here's Chris' super-duper introduction to sea-ice. Hopefully, I'll find the time to go into a bit more detail in a later post. What the hell is sea ice? It's probably a good idea to spend 5 seconds defining my terms and say, clearly, what sea ice is an what it 'ain't, as this question seems to trip-up some of the more gullible climate-change skeptics, including politicians and journalists with a far greater readership and influence than myself. Sea ice isn't just "ice in the ocean". Sea ice is ice that forms when ocean water - that is water that is already in the ocean - freezes. When ocean water freezes, the total amount of water in the ocean-ice system stays the same. Thus melting sea-ice cannot contribute to the rise in sea-levels expected to occur (and already being observed) with global warming. Sea-ice is a different kettle-of-fish from land ice, such as the ice in glaciers and ice shelves. Icebergs, for example, are formed by chunks of ice falling of iceshelves or glaciers. Icebergs, although being ice in the sea, are not sea ice. Clear as mud, n'est-ce pas? When ice that was on land either melts and flows into the ocean, or breaks off and falls into the ocean the total amount of water in the ocean increases. In this way, melting glaciers and collapsing ice-shelves can contribute to sea-level rise. That's not to say that melting sea ice is unimportant for the climate system. Au contraire mes ami(e)s, sea-ice has extremely large influence on the climate, which I'll go into in a moment. But those effects don't, directly at least, lead to sea-level rise. Where, and why, does the ocean freeze? 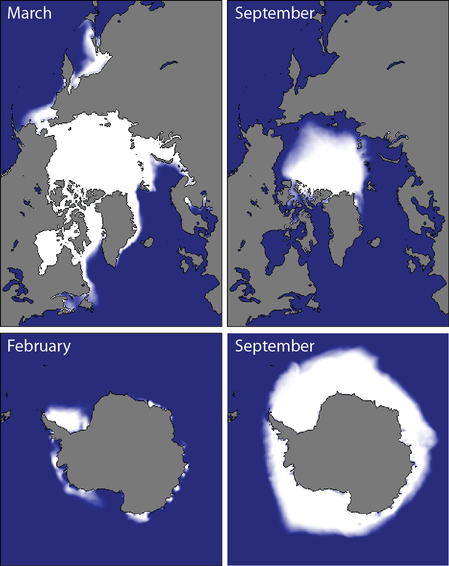 The extent of sea-ice in a "normal" year for the northern (top) and southern (bottom) hemispheres. Taken from the National Snow and Ice Data Centre (http://nsidc.org/cryosphere/sotc/sea_ice.html) The extent of sea-ice in a "normal" year for the northern (top) and southern (bottom) hemispheres. Taken from the National Snow and Ice Data Centre (http://nsidc.org/cryosphere/sotc/sea_ice.html) Since sea ice occurs when the ocean freezes, a better question is probably "why doesn't the ocean freeze more?" After all, much of the ocean is below 0 degrees Celsius, and as someone who has spent the better part of two weeks have this friggin' freezing water poured all over my hands while taking water samples, I can assure you it remains very liquid (it also remains very cold). In fact, sea ice is remarkable because it is rare: only about 10 percent of the ocean surface freezes regularly, and the vast majority of that occurs poleward of about 60 degrees in the Arctic or Antarctic seas. Sea-ice really only becomes bothersome for shipping in northern latitudes and mostly in the winter (there is not much shipping in the regions in the southern hemisphere where sea-ice forms, so the only people who are bothered by it are us scientists and cruise ship patrons who have shelled out exorbitant sums of cash to visit <insert exotic-sounding Antarctic local here> and can't because it's covered by 5 meters of heavy pack-ice). From where I'm sitting at the moment, the water temperature at the surface is -1.4 degrees C. Why isn't the ocean here frozen solid? Well, the short answer is "salinity". The long answer is also "salinity". Essentially, the presence of salt in water lowers its freezing temperature. The freezing temperature of "normal" seawater is about -1.9 degrees. However, the more salt, the lower the freezing temperature. In the Weddell Sea, where I currently find myself, water freezes at about -3 degrees. However, some important shipping areas, such as the Baltic Sea, in between the Scandinavian countries and the rest of Europe, has a much lower salinity and freezes up at a higher water temperature. It should be pretty unsurprising that sea-ice varies with the seasons. It grows in winter and retreats in summer. However, just as one summer may be warmer than another summer, sea-ice shows strong inter-annual variability. That's because sea-ice growth is affected by many external factors: temperature (duh...), but also winds (which not only cool the ocean surface but also blow sea-ice around in complicated ways), waves (which break-up the ice - ice, in turn, kills waves), snow cover which insulates ice and helps prevent it from melting, the amount of sun beating down on the ice surface and a whole bunch of other stuff. The upshot is that a colder year does not necessarily equal a greater ice extent, simply because the wind, for example, may blow the ice back to where it came from. The physics of sea-ice are immensely complicated. Ice is constantly bashing into other ice, breaking, melding together, melting, dissolving and reforming, all of which make it a great challenge to model. Why should you care about sea-ice? Unless you are a Russian freighter captain trying to pilot a cargo of 857,248 beanie-babies (beanie-babies Chris? Beanie-babies? Seriously, what the hell man? Who has given a single solitary thought to beanie-babies in the last 20 years?) through the sea of Okhotsk in winter, sea-ice isn't likely to affect your day-to-day activities. But we oceanographers and climatologists are interested in sea-ice as it has extremely important effects on the climate system. This is a subject in and of itself, and impossible to encapsulate in a single blog post. As I keep mentioning, I'll try to fill in the gaps in a dedicated post. Probably the most well known of these effects is the "ice albedo" effect. Simply put, ocean water, absorbs most of the sunlight that falls on it, whereas ice, and particularly snow covered ice, reflects pretty much all of it. As such, sea ice acts as kind of inbuilt cooling mechanism for the planet. However, sea-ice has a multitude of other effects on the climate, both in the local region where it is formed, and for the global climate. One that does not get a lot of press, even in scientific circles, is that sea-ice puts a cap over the ocean where it forms, which means that the ocean is now cut-off from the atmosphere. Under normal (that is, open ocean) conditions, the atmosphere and the ocean exchange heat, mass and momentum. Winds, blowing over the ocean's surface exert a stress which is basically a transfer of momentum from the atmosphere to the ocean. Similarly, warm or cold air blowing over the ocean surface can either heat-up or cool down the ocean water. Both of these phenomena are essentially what drive the major ocean current systems. When sea-ice is present, however, winds blow not directly on the water but on the ice itself. Now the atmosphere must talk to the ocean via an intermediary, the ice. Wind blows on the ice, the ice moves, and the moving ice pushing on the water causes the water to move, and so-on.The ice also insulates the ocean, like a lid, and stops it from losing heat to the atmosphere. Somewhat counter-intuitively, sea-ice helps keep the ocean warm (if you can call -1.5 degrees warm). Sea ice has some other important effects as well. One of the most well studied is the phenomena of "brine rejection". Sea ice, despite being made-up of ocean water, is very fresh: there is almost no salt in sea ice. When the ice crystals form, they kick out the dissolved salt. That salt has to go somewhere, so it generally goes directly into the ocean below, making it more salty and, therefore, denser. The formation of sea-ice is very important for the formation of the very dense Antarctic Bottom Water, that fills much of the ocean from the bottom-up (I've written a little about AABW before). If there were no sea-ice, the interior of the oceans would look very, very different. Finally, the area near the edge of the sea ice, the so-called "Marginal Ice Zone" is incredibly productive biologically. In these regions, a complex interaction between ocean, wind and the ice, cause nutrients (read: fish poo. Lots and lots of fish poo) to be brought to the surface from below, where it is used by microscopic water-born plants (pytoplankton) who get eaten by tiny animals (zooplankton), who get eaten by bigger critters, and so on up the food chain. Primary productivity, a measure of the intensity of the base of the food chain, is off the charts in these regions, which has all sorts of flow on effects, from sucking of CO2 out of the atmosphere (pytoplankton, being plants, consume CO2 during photosynthesis) to the colour of the ocean. The list of ways that sea-ice influences the global climate doesn't end here, although my condensed version does. A more detailed post is in the making. Measuring sea-iceWhen we talk about measuring sea-ice, we're generally interested in four things, which are, in order of importance:
Let me illustrate my point with an example. Take a look at this photo:  To explain the bright yellow hood, I'll simply state that it was -21 degrees when this photo was taken. To explain the bright yellow hood, I'll simply state that it was -21 degrees when this photo was taken. Now, tell me what the concentration of the ice is, and how thick it is. The photo shows a bit of a mess. Ice bergs (which, I'll remind you, are not sea-ice) mixed in with snow (also not sea ice) mixed with cracks in the ice, mixed with man-eating penguins, mixed with who knows what else. It takes a skilled observer and a large amount of experience to make useful observations of ice concentrations and types from ships. Measuring thickness is even more difficult. Much like the stereotypical iceberg, where the majority of a chunk of sea ice lies below the surface of the water (fun fact: the part above the surface is called the 'freeboard', the part below, the 'draft'. You are now primed to crush your next local pub quizz-night). Thickness is vitally important for shipping. The James Clarke Ross can break ice that's one meter thick with no problems. Ice that's 5 meters thick can break the James Clarke Ross with no problems. So to measure ice-thickness, we use a sophisticated instrument called a Statistical Transmorphic Ice Calibration Kit (STICK). Here's your humble author brandishing this piece of cutting edge technology. Now, here is a photo of STICK in action.  Flying a quad-copter over Antarctica. Sometimes, I think my job is pretty cool. Flying a quad-copter over Antarctica. Sometimes, I think my job is pretty cool. Yes, that's right. We use a broom handle, divided up into 10cm sections, to measure the thickness of ice turned over by the ship as it passes through an ice field. Sophisticate, eh? Sometimes, I've waited 15-20 minutes for a nice block of ice to turn over. Which, in Antarctic temperatures (it's been consistently below minus 15 degrees C), starts to wear at ones patience. We've also recently been experimenting with using a drone to estimate the sea-ice concentrations, types and floe sizes, although to-date, we haven't had much luck as our opto-electrical (visible) camera failed. But in the future drone technology will allow us to combined ship based estimates with quick-n-easy aerial surveys, something I'm really looking forward to working with more in the future, and not just because, like any self-respecting nerd, I like playing with shiny new toys. I'm going to finish up this post here, as it's already gotten longer than I had initially planned. Suffice to say sea ice is both super important, and super interesting. Hopefully, I've given you a taste of that. A note on attribution Many of these photos weren't taken by me. In fact, I've been stealing photos left, right and centre, trying to get the best ones, for your viewing pleasure.
Many in this post were take by Helen, the ship's doctor. She blogs on our adventures at Baby it's Cold Down Here and has some amazing photos. So go show her some love. Many other photos come from my friends and colleagues, Yves and Sara, our resident biologists who do ice observations while I sleep. They have their own blog (en francais, bien sur. On bosse en France normalement!) over at Immersion sous la Banquise en Antarctique (Immersion under the Antarctic Sea Ice). More fantastic photos await.
1 Commentaire
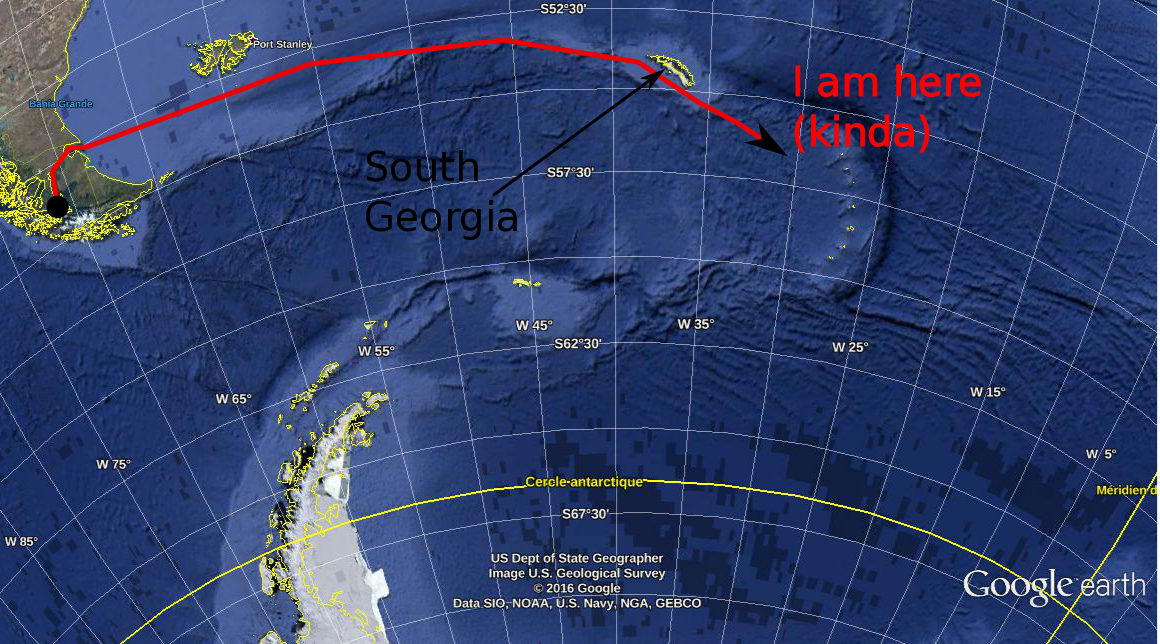
In my last post, I explained a little about why we oceanographers are interested in the temperature (T) and salinity (S) of the oceans. Today, I'll show you how we do it from ocean going research ships, illustrating with a couple of photos from the first couple of measurements we made south of the island of South Georgia (one of the many places that Ernest Shackleton didn't die). How it's done. This is a CTD. CTD stands for Conductivity, Temperature, Depth. It's not just a clever name: this device really does measure the conductivity, the temperature and the depth. Oceanographers are known for their creative naming conventions. Conductivity is a measure of how electrically conductive a substance is (if you've ever done electronics, you'll know conductivity as the inverse resistance). Water that is more saline is more electrically conductive. With careful calibration of the instrument - a job in an of itself - if we know the conductivity of a water sample, we know its salinity. From afar, the CTD might look like a rack of bottles. That's because a CTD is, primarily, a rack of bottles. Ours has 24 bottles that can hold 12 litres of water. These bottles are attached by latex wires to a magnetically operated trigger. By lowering the CTD into the ocean on a winch, we can take water samples at any depth that interests us. An operator, usually a skilled and experienced oceanographer, but in some cases, me, "fires" a bottle, which causes it to snap closed at a depth that interests us. We've got 24 bottles, which means we could, if we wanted to, sample water from 24 different depths. However, for a lot of chemistry and biology work (measuring, for example, the amount of CO2 or oxygen dissolved in the water, or the amount of plankton or whatever), you need a lot of water, so we'll often "fire" two bottles at the same depth to give our colleagues more material to work with. Here's a CTD on wire being winched over the side of the ship. 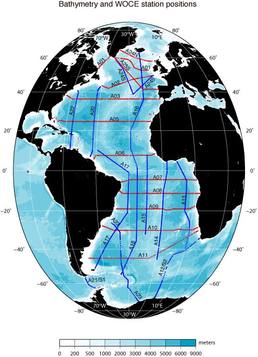 WOCE sections in the Atlantic (from the WOCE Atlas woceatlas.ucsd.edu/) WOCE sections in the Atlantic (from the WOCE Atlas woceatlas.ucsd.edu/) We lower the CTD down to about 10 meters above the ocean floor (getting the CTD stuck on the bottom of the ocean is generally considered to be a dick move). We then winch it up to the surface, taking water samples as we go. When we get it back on the ship, the water samples are divided up among the assembled waiting chemists, who then proceed to science the crap out of them. Some of the water samples are scienced in labs on board the ship. Others are packed up and sent back home at the end of the campaign for further sciencing on land. Some lab equipment, such as electron microscopes, and... I dunno... an elephant or something (I'm not a chemist) you just can't take on a boat. These days, CTDs also have electronic temperature and conductivity (salinity) probes which measure, in real time, the T and S of the ocean, as well as some other interesting quantities such as "florescence" (how much ocean water glows when you shine a light on it... an indirect measure of the amount of phytoplankton in the water) or dissolved oxygen. The information is the sent back up to the ship via a cable and we can monitor how the T & S change with depth as the CTD is lowered and raised. This real time information helps us decided at exactly which depth we want to sample water at. Most plankton activity, for example, happens relatively close to the surface in an area called the "mixed layer" whose depth depends on the strength of the wind, waves, location, time of year, etc... The mixed layer gives itself away by being ... well... mixed - that is, its temperature and salinity are relatively constant throughout its depth. The bottom of the mixed layer is marked by a very strong temperature gradient. For our friends the biologists, we want to be very sure to sample this region, so we keep a close eye on the temperature as the CTD goes down (and comes up again) to give us an idea about where in the water column to sample. The electronic temperature and conductivity probes are good enough these days that we use their data pretty much as we would bottle samples. However, we do check the temperature and salinity collected via the bottle samples against the electronic equipment to make such that both are working well, and that the calibrations haven't "drifted" too much. Where do we do it? The oceans are big. Boats are small. Even with 10,000 boats piloted by crack team of super intelligent talking mice, covering the world's oceans would be a futile task. That's why, back in the late 80's, a scheme was launched, called the World Ocean Circulation Experiment (WOCE) to divide the ocean's up into "sections" or "transects". These sections were carefully chosen because the people in charge of WOCE wanted to measure certain interesting ocean phenomena. For example, the section SR1, which runs between the tip of South America and the Antarctic Peninsula was designed to measure the strength of the Antarctic Circumpolar Current as it passes through Drake Passage (the passage between, err... the tip of South America and the Antarctic Peninsula). Each section is divided into a number of "stations": points along the line where we deploy our CTD (and other instruments if you've got 'em). The ship comes to a stop at each of the stations, and we role out the ol' CTD and take a profile at that location. Ocean going field campaigns should, subject to weather conditions, timing, cruise objectives, etc... take their measurements along one of these "sections" at its appropriate stations. By repeatedly sampling the same parts of the ocean, we can build up a good body of knowledge about the ocean in particular areas, as well as see how things change in that area with time. Systematic sampling is usually (although not always) better than sampling au hazard. It enables easy comparison with what's gone on before and allows (kinda) continuous monitoring of certain interesting phenomena. Before we get to the Weddell Sea, we on the WAPITI cruise will be "occupying" (that is, re-sampling) the southern part of the A23 line - between the bottom of South Georgia island and the Antarctic continent. A23 is a bit of a "forgotten" transect, having only been occupied a few times previously. As it's on the way to our principle destination, we (and by "we" I mean my boss and the head-honchos at the British Antarctic Survey) thought that it would be a good idea to take some samples along this transect during the voyage. So that's the basics of how we measure T & S (and some other stuff) during a research cruise. I should mention that this isn't the only way to measure T & S. We can also measure both quantities by satellites (but only at the surface, of course) by moored instruments stuck to the ocean floor (but on in one place, of course) or by drifting robots called Argo floats (but then you don't know where exactly you'll measure).
In my next post I'll describe the hows and whys of measuring the speed of ocean currents and why that's an important thing to do. |
Chris Chapman
I'm a physical oceanographer at LOCEAN Paris. My research concentrates on the Southern Ocean and its role in the climate system. ArchivesCategories |
Proudly powered by Weebly






 Flux RSS
Flux RSS